Navigation :
(Acyl)polyamines
-
2,4-(OH)₂-PhAc
-
2,5-(OH)₂-Bz
-
4-OH-Bz
-
4-OH-IndAc
-
4-OH-PhAc
-
4-OH-PhLac
-
Ac
-
Bz
-
IndAc
-- IndAc3(Me)43
-- IndAc3(Me₂)4(Me)3(NMe)⁺
-- IndAc3(Me₂)4(Me₂)3Arg²⁺
-- IndAc3(OH)3
-- IndAc3(OH)3(OH)33
-- IndAc3(OH)3(OH)34
-- IndAc3(OH)3(OH)343
-- IndAc3(OH)3(OH)35(NMe₃)⁺
-- IndAc3(OH)3(OH)43
-- IndAc3(OH)33
-- IndAc3(OH)333
-- IndAc3(OH)333(OH)3
-- IndAc3(OH)3334Gu
-- IndAc3(OH)3335(NMe₃)⁺
-- IndAc3(OH)334
-- IndAc3(OH)334(NMe)
-- IndAc3(OH)3343
-- IndAc3(OH)33433
-- IndAc3(OH)3343Ac
-- IndAc3(OH)334Gu
-- IndAc3(OH)335
-- IndAc3(OH)335(NMe)
-- IndAc3(OH)335(NMe₂)
-- IndAc3(OH)335(NMe₃)⁺
-- IndAc3(OH)343
-- IndAc3(OH)35(NMe₃)⁺
-- IndAc3(OH)4
-- IndAc3(OH)43
-- IndAc33(OH)33
-- IndAc333
-- IndAc3333
-- IndAc3334
-- IndAc33343
-- IndAc3334Gu
-- IndAc3335(NMe₃)⁺
-- IndAc334
-- IndAc3343
-- IndAc335(NMe₃)⁺
-- IndAc34
-- IndAc34(OH)33
-- IndAc343
-- IndAc3433
-- IndAc343Ac
-- IndAc35(NMe₃)⁺
-- IndAc353
-- IndAc4
-- IndAc4(OH)3
-- IndAc4(OH)3(OH)3
-- IndAc4(OH)3(OH)33
-- IndAc4(OH)3(OH)333
-- IndAc4(OH)33
-- IndAc4(OH)333
-- IndAc4(OH)333Ac
-- IndAc43
-- IndAc43(OH)33
-- IndAc433
-- IndAc4333
-- IndAc4333Ac
-- IndAc5(OH)3(OH)33
-- IndAc5(OH)33
-- IndAc533
-- IndAc5333
-- IndAcAsn3(Me)43
-- IndAcAsn343
-- IndAcAsn353
-- IndAcAsn4
-- IndAcAsn43
-- IndAcAsn433
-- IndAcAsn433ßAla4
-- IndAcAsn433ßAla4ßAla4
-- IndAcAsn4Lys(Me₂)3Arg⁺
-- IndAcAsn4Lys(Me₂)3⁺
-- IndAcAsn5
-- IndAcAsn53
-- IndAcAsn533
-- IndAcAsn533Arg
-- IndAcAsn5Gly4
-- IndAcAsn5Gly4ßAla4
-- IndAcAsn5Gly4ßAla4ßAla4
-- IndAcAsn5Gly4ßAla4ßAla4ßAla4
-- IndAcAsn5Lys(Me)3Arg
-- IndAcAsn5Lys(Me₂)3Arg⁺
-- IndAcAsn5Orn
-- IndAcAsn5OrnArg
-- IndAcAsn5ßAla3Arg
-- IndAcAsn5ßAla3ßAla4ßAla4
-- IndAcAsn5ßAla3ßAla4ßAla4ßAla4
-- IndAcAsn5ßAla4
-- IndAcAsn5ßAla43
-- IndAcAsn5ßAla43ßAla4
-- IndAcAsn5ßAla43ßAla4ßAla4
-- IndAcAsn5ßAla4Arg
-- IndAcAsn5ßAla4ßAla4
-- IndAcAsn5ßAla4ßAla4ßAla4
-- IndAcAsn5ßAla4ßAla4ßAla4ßAla4
-- IndAcGln4
-- IndAcOrnAsn5ßAla4ßAla4ßAla4
-- IndAcOrnAsp5ßAla4ßAla4
-- IndAcßAla343
-- IndAcßAla353
-
IndLac
-
OH-OMe-Bz
-
PhAc
-
Phe
-
PhLac
-
Prop
-
Trp
-
Tyr
-
Unknown acylpolyamines
-
Free polyamines (PA)
Small compounds
Analytical tools
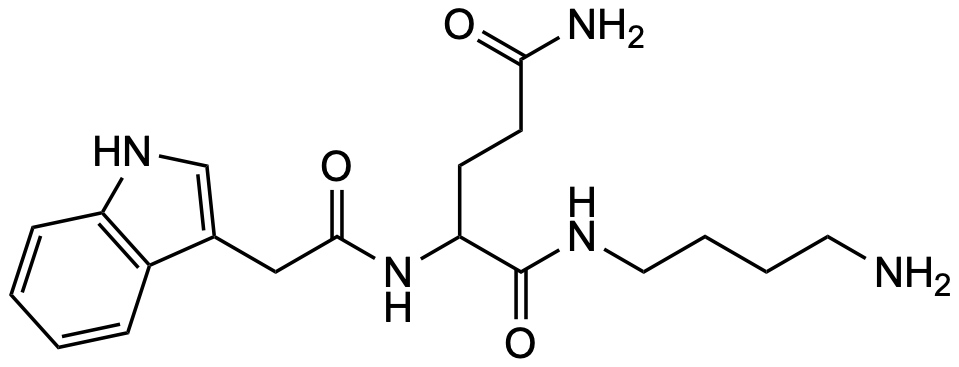
Contact us IndAcGln4
General Description Name Value Level S-3 / C-1 Discovered 2020 / P. bistriata Synonym — Molecular formula C₁₉H₂₇N₅O₃ CAS — SMILES O=C(NC(CCC(N)=O)C(NCCCCN)=O)CC1=CNC2=C1C=CC=C2InChI InChI=1S/C19H27N5O3/c20-9-3-4-10-22-19(27)16(7-8-17(21)25)24-18(26)11-13-12-23-15-6-2-1-5-14(13)15/h1-2,5-6,12,16,23H,3-4,7-11,20H2,(H2,21,25)(H,22,27)(H,24,26)Precursor 1 [M+H]⁺ 374.21867 Precursor 2 [M+2H]²⁺ 187.61297 Precursor 3 HDX 7 Precursor HDX 1 [M(D₇)+D]⁺ 382.26888 Precursor HDX 2 [M(D₇)+2D]²⁺ 192.14122 Precursor HDX 3 Rt 10.76 Rt HDX 9.55
Calculated MS/MS fragments # a b c ta z y tz 1 286.11862 268.10805 269.09207 303.14517 72.08078 55.05423 89.10732 2 357.19212 339.18155 340.16557 374.21867 200.13935 183.11280 217.16590
Additional MS/MS fragments m/z Annotation 130.06513 a’ 158.06004 a0
Recorded MS/MS spectra pdf Precursor Co-eluting Spider Source Author Data 374.21867 P. bistriata Prof. Dr. Wagner Ferreira dos Santos, BRA Y. M. Forster Data HDX P. bistriata Prof. Dr. Wagner Ferreira dos Santos, BRA Y. M. Forster
References Title Reference Spider Name Content Link Elucidation of the structure and synthesis of neuroprotective low molecular mass components of the Parawixia bistriata spider venom Y. M. Forster, J. L. Green, A. Khatiwada, J. L. Liberato, P. A. Narayana Reddy, J. M. Salvino, S. Bienz, L. Bigler, W. Ferreira dos Santos, A. C. K. Fontana, ACS Chem Neurosci., 2020 P. bistriata ESI-MS/MS Link Low molecular mass compounds in spider venom Y. M. Forster, S. Bienz, L. Bigler, 2020, in preparation div. Link
Spider species Spider species Family Discovered Parawixia bistriata Araneidae 2020 / Y. M. Forster