IndAc4333

General Description
| Name | Value |
|---|---|
| Level | S-1 / C-1 |
| Discovered | 1991 / H. curta |
| Synonym | AG 416b / HO 416b |
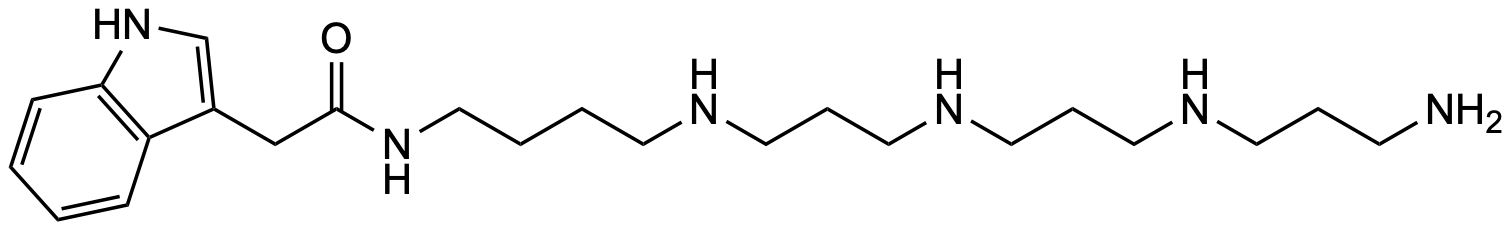
| Molecular formula | C₂₃H₄₀N₆O |
| CAS | 133823-88-8 |
| SMILES | O=C(NCCCCNCCCNCCCNCCCN)CC1=CNC2=C1C=CC=C2 |
| InChI | InChI=1S/C23H40N6O/c24-10-5-12-26-14-7-16-27-15-6-13-25-11-3-4-17-28-23(30)18-20-19-29-22-9-2-1-8-21(20)22/h1-2,8-9,19,25-27,29H,3-7,10-18,24H2,(H,28,30) |
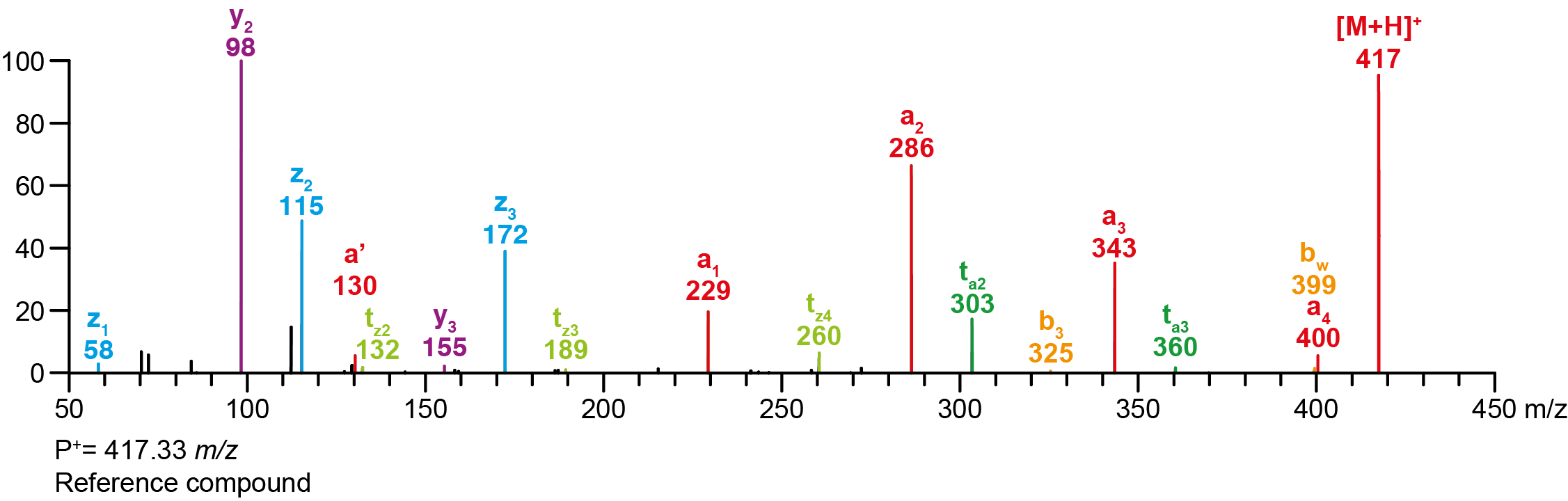
| Precursor 1 [M+H]⁺ | 417.33364 |
| Precursor 2 [M+2H]²⁺ | 209.17046 |
| Precursor 3 | |
| HDX | 7 |
| Precursor HDX 1 [M(D₇)+D]⁺ | 425.38385 |
| Precursor HDX 2 [M(D₇)+2D]²⁺ | 213.69870 |
| Precursor HDX 3 | |
| Rt | 9.00 |
| Rt HDX | 7.32 |
Calculated MS/MS fragments
| # | a | b | c | ta | z | y | tz |
|---|---|---|---|---|---|---|---|
| 1 | 229.13354 | 211.12297 | 212.10699 | 246.16009 | 58.06513 | 41.03858 | 75.09167 |
| 2 | 286.19139 | 268.18082 | 269.16484 | 303.21794 | 115.12297 | 98.09643 | 132.14952 |
| 3 | 343.24924 | 325.23867 | 326.22269 | 360.27579 | 172.18082 | 155.15428 | 189.20737 |
| 4 | 400.30709 | 382.29652 | 383.28054 | 417.33364 | 243.25432 | 226.22777 | 260.28087 |
Additional MS/MS fragments
| m/z | Annotation |
|---|---|
| 130.06513 | a’ |
| 158.06004 | a0 |
Recorded MS/MS spectra
| Precursor | Co-eluting | Spider | Source | Author | |
|---|---|---|---|---|---|
| Data | 417.33364 | synth. IndAc4333 | UZH Bienz lab, CHE | Y. M. Forster | |
| Data | 209.17046 | synth. IndAc4333 | UZH Bienz lab, CHE | Y. M. Forster | |
| Data | 417.33364 | A. aperta | Fauna Laboratories Ltd., KAZ | Y. M. Forster | |
| Data | 209.17046 | A. aperta | Fauna Laboratories Ltd., KAZ | Y. M. Forster | |
| Data | HDX | A. aperta | Fauna Laboratories Ltd., KAZ | Y. M. Forster | |
| Data | 417.33364 | A. potteri | Fauna Laboratories Ltd., KAZ | Y. M. Forster | |
| Data | 209.17046 | A. potteri | Fauna Laboratories Ltd., KAZ | Y. M. Forster | |
| Data | HDX | A. potteri | Fauna Laboratories Ltd., KAZ | Y. M. Forster | |
| Data | 417.33364 | H. curta | Fauna Laboratories Ltd., KAZ | Y. M. Forster | |
| Data | 209.17046 | H. curta | Fauna Laboratories Ltd., KAZ | Y. M. Forster | |
| Data | HDX | H. curta | Fauna Laboratories Ltd., KAZ | Y. M. Forster | |
| Data | 417.33364 | Hololena sp. | Spider Pharm, USA | Y. M. Forster | |
| Data | 209.17046 | Hololena sp. | Spider Pharm, USA | Y. M. Forster | |
| Data | HDX | Hololena sp. | Spider Pharm, USA | Y. M. Forster |
References
| Title | Reference | Spider | Name | Content | Link |
|---|---|---|---|---|---|
| Paralytic and insecticidal toxins from the funnel web spider, Hololena curta | G. B. Quistad, C. C. Reuter, W. S. Skinner, P. A. Dennis, S. Suwanrumpha, E. W. Fu, Toxicon 1991, 29, 329-336 | H. curta | HO 416b | FAB-MS/MS (ns), Activity-studies | Link |
| Polyamine toxins from spiders and wasps | A. Schäfer, H. Benz, W. Fiedler, A. Guggisberg, S. Bienz, M. Hesse, The Alkaloids 1994, 45, 1-125 | H. curta | HO 416b | Review | Link |
| Total synthesis of polyamine toxin Ho-416b utilizing the 2-nitrobenzenesulfonamide protecting group | Y. Hidai, T. Kan, T. Fukuyama, Tetrahedron Letters 1999, 40, 4711-4714 | HO 416b (1) | Synthesis, NMR (ns), FAB-MS/MS (ns) | Link | |
| Acylpolyamines: Mass spectrometric analytical methods for Araneidae spider acylpolyamines | Y. Itagaki , T. Nakajima , Toxin Rev. 2000, 19, 23-52 | H. curta | HO 416b | Review | Link |
| Total synthesis of polyamine toxin HO-416b and Agel-489 using a 2-nitrobenzenesulfonamide strategy | Y. Hidai, T. Kan, T. Fukuyama, Chem. Pharm. Bull 2000, 48, 1570-1576 | HO 416b (1) | Synthesis, NMR, FAB-MS/MS | Link | |
| Solid phase syntheses of polyamine toxins Ho-416b and PhTx-433. Use of an efficent polyamide reduction strategy that facilitates access to branched analogues | F. Wang, S. Manku, D. G. Hall, Organic Letters 2000, 2, 1581-1583 | HO 416b (1) | Synthesis | Link | |
| The acylpolyamines from the venom of the spider Agelenopsis aperta | S. Chesnov, L. Bigler, M. Hesse, Helv. Chim. Acta 2001, 84, 2178-2197 | A. aperta | AG 416b | APCI-MS/MS | Link |
| Detection and characterization of natural polyamines by high-performance liquid chromatography-atmospheric pressure chemical ionization (electrospray ionization) mass spectroscopy | S. Chesnov, L. Bigler, M. Hesse, Eur. J. Mass Spectrom. 2002, 8, 1-16 | A. aperta | AG 416b | Review | Link |
| Solid-phase synthesis of polyamine spider toxins and correlation with natural products by HPLC-MS/MS | N. Manov, M. Tzouros, S. Chesnov, L. Bigler, S. Bienz, Helv. Chim. Acta 2002, 85, 2827-2846 | A. aperta | Synthesis, NMR, APCI-MS/MS | Link | |
| Tandem mass spectrometric investigation of acylpolyamines of spider venoms and their 15N-labeled derivatives | M. Tzouros, N. Manov, S. Bienz, L. Bigler, J. Am. Soc. Mass Spectrom. 2004, 15, 1636-1643 | APCI-MS/MS | Link | ||
| Low molecular mass compounds in spider venom | Y. M. Forster, S. Bienz, L. Bigler, 2020, in preparation | div. | Link |
Spider species
| Spider species | Family | Discovered |
|---|---|---|
| Agelenopsis aperta | Agelenidae | 2001 / S. Chesnov |
| Agelenopsis potteri | Agelenidae | 2020 / Y. M. Forster |
| Hololena curta | Agelenidae | 1991 / G. B. Quistad |
| Hololena sp. | Agelenidae | 2020 / Y. M. Forster |