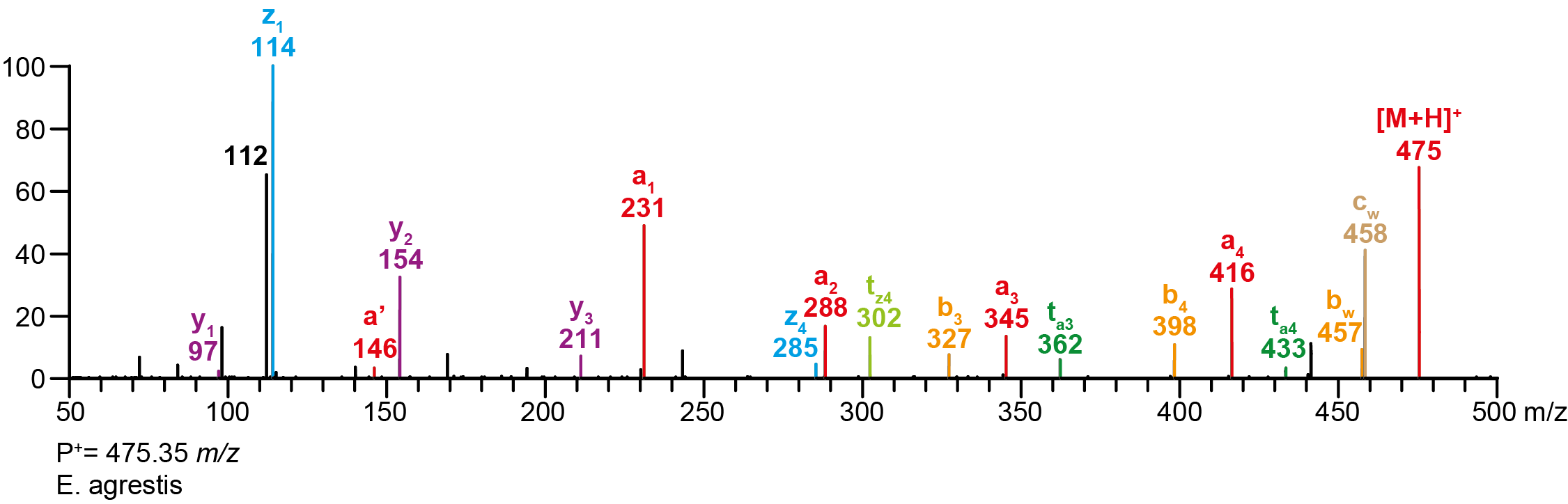Navigation :
(Acyl)polyamines
-
2,4-(OH)₂-PhAc
-
2,5-(OH)₂-Bz
-
4-OH-Bz
-
4-OH-IndAc
-- 4-OH-IndAc3
-- 4-OH-IndAc3(Me₂)4(Me)3(NMe)⁺
-- 4-OH-IndAc3(Me₂)4(Me)3Arg⁺
-- 4-OH-IndAc3(Me₂)4(Me₂)3Arg²⁺
-- 4-OH-IndAc3(Me₂)4(NMe)⁺
-- 4-OH-IndAc3(Me₂)4(NMe₂)⁺
-- 4-OH-IndAc3(OH)3
-- 4-OH-IndAc3(OH)3(OH)33
-- 4-OH-IndAc3(OH)3(OH)34
-- 4-OH-IndAc3(OH)3(OH)343
-- 4-OH-IndAc3(OH)3(OH)35(NMe₃)⁺
-- 4-OH-IndAc3(OH)3(OH)43
-- 4-OH-IndAc3(OH)33
-- 4-OH-IndAc3(OH)333
-- 4-OH-IndAc3(OH)3334Gu
-- 4-OH-IndAc3(OH)334
-- 4-OH-IndAc3(OH)3343
-- 4-OH-IndAc3(OH)3343Ac
-- 4-OH-IndAc3(OH)334Gu
-- 4-OH-IndAc3(OH)335
-- 4-OH-IndAc3(OH)335(NMe)
-- 4-OH-IndAc3(OH)335(NMe₂)
-- 4-OH-IndAc3(OH)335(NMe₃)⁺
-- 4-OH-IndAc3(OH)34
-- 4-OH-IndAc3(OH)343
-- 4-OH-IndAc3(OH)34Gu
-- 4-OH-IndAc3(OH)35(NMe₃)⁺
-- 4-OH-IndAc3(OH)4
-- 4-OH-IndAc3(OH)43
-- 4-OH-IndAc3333
-- 4-OH-IndAc33334Gu
-- 4-OH-IndAc3334
-- 4-OH-IndAc33343
-- 4-OH-IndAc3334Gu
-- 4-OH-IndAc3335(NMe₃)⁺
-- 4-OH-IndAc334
-- 4-OH-IndAc3343
-- 4-OH-IndAc334Gu
-- 4-OH-IndAc335(NMe₃)⁺
-- 4-OH-IndAc34
-- 4-OH-IndAc343
-- 4-OH-IndAc3433
-- 4-OH-IndAc35
-- 4-OH-IndAc353
-- 4-OH-IndAc4(OH)3
-- 4-OH-IndAc4(OH)3(OH)33
-- 4-OH-IndAc4(OH)33
-- 4-OH-IndAc4(OH)333
-- 4-OH-IndAc4(OH)3333
-- 4-OH-IndAc43
-- 4-OH-IndAc433
-- 4-OH-IndAc4333
-- 4-OH-IndAc4Gu
-- 4-OH-IndAc5
-- 4-OH-IndAc5(NMe₃)⁺
-- 4-OH-IndAc5(OH)33
-- 4-OH-IndAc5(OH)3333
-- 4-OH-IndAc53
-- 4-OH-IndAc533
-- 4-OH-IndAc5333
-- 4-OH-IndAcAsn3(Me)4(Me)3Arg
-- 4-OH-IndAcAsn3(Me)43
-- 4-OH-IndAcAsn333
-- 4-OH-IndAcAsn34
-- 4-OH-IndAcAsn343
-- 4-OH-IndAcAsn34ßAla3
-- 4-OH-IndAcAsn34ßAla4
-- 4-OH-IndAcAsn353
-- 4-OH-IndAcAsn4
-- 4-OH-IndAcAsn4(Me)3
-- 4-OH-IndAcAsn43
-- 4-OH-IndAcAsn433
-- 4-OH-IndAcAsn433Arg
-- 4-OH-IndAcAsn433Lys
-- 4-OH-IndAcAsn433ßAla4
-- 4-OH-IndAcAsn433ßAla4ßAla4
-- 4-OH-IndAcAsn43Arg
-- 4-OH-IndAcAsn43Lys
-- 4-OH-IndAcAsn43ßAla4
-- 4-OH-IndAcAsn4Lys(Me₂)3Arg⁺
-- 4-OH-IndAcAsn4ßAla3Arg
-- 4-OH-IndAcAsn4ßAla3ßAla4ßAla4
-- 4-OH-IndAcAsn4ßAla3ßAla4ßAla4ßAla4
-- 4-OH-IndAcAsn4ßAla4
-- 4-OH-IndAcAsn4ßAla43
-- 4-OH-IndAcAsn4ßAla4Arg
-- 4-OH-IndAcAsn4ßAla4ßAla4
-- 4-OH-IndAcAsn5
-- 4-OH-IndAcAsn53
-- 4-OH-IndAcAsn533
-- 4-OH-IndAcAsn533Arg
-- 4-OH-IndAcAsn5Gly4
-- 4-OH-IndAcAsn5Gly4ßAla4
-- 4-OH-IndAcAsn5Gly4ßAla4ßAla4
-- 4-OH-IndAcAsn5Gly4ßAla4ßAla4ßAla4
-- 4-OH-IndAcAsn5Lys(Me)3Arg
-- 4-OH-IndAcAsn5Lys(Me₂)3Arg⁺
-- 4-OH-IndAcAsn5ßAla3
-- 4-OH-IndAcAsn5ßAla3Arg
-- 4-OH-IndAcAsn5ßAla3ßAla4ßAla4
-- 4-OH-IndAcAsn5ßAla3ßAla4ßAla4ßAla4
-- 4-OH-IndAcAsn5ßAla4
-- 4-OH-IndAcAsn5ßAla43
-- 4-OH-IndAcAsn5ßAla43Arg
-- 4-OH-IndAcAsn5ßAla43ßAla4
-- 4-OH-IndAcAsn5ßAla43ßAla4ßAla4
-- 4-OH-IndAcAsn5ßAla4Arg
-- 4-OH-IndAcAsn5ßAla4OrnArg
-- 4-OH-IndAcAsn5ßAla4ßAla4
-- 4-OH-IndAcAsn5ßAla4ßAla4ßAla4
-- 4-OH-IndAcAsn5ßAla4ßAla4ßAla4ßAla4
-- 4-OH-IndAcLysMe53Arg
-- 4-OH-IndAcLysMe₂5(Me)3Arg
-- 4-OH-IndAcOrnAsn5ßAla4ßAla4
-- 4-OH-IndAcOrnAsn5ßAla4ßAla4ßAla4
-- 4-OH-IndAcßAla343
-- 4-OH-IndAcßAla353
-
4-OH-PhAc
-
4-OH-PhLac
-
Ac
-
Bz
-
IndAc
-
IndLac
-
OH-OMe-Bz
-
PhAc
-
Phe
-
PhLac
-
Prop
-
Trp
-
Tyr
-
Unknown acylpolyamines
-
Free polyamines (PA)
Small compounds
Analytical tools
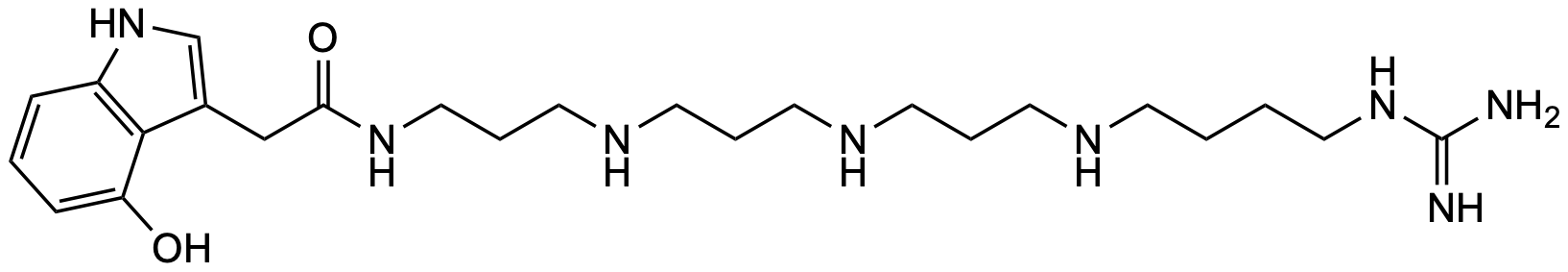
Contact us 4-OH-IndAc3334Gu
General Description Name Value Level S-3 / C-1 Discovered 2002 / P. luctuosa Synonym PB 474 Molecular formula C₂₄H₄₂N₈O₂ CAS — SMILES NC(NCCCCNCCCNCCCNCCCNC(CC1=CNC2=C1C(O)=CC=C2)=O)=NInChI InChI=1S/C24H42N8O2/c25-24(26)31-15-2-1-9-27-10-4-11-28-12-5-13-29-14-6-16-30-22(34)17-19-18-32-20-7-3-8-21(33)23(19)20/h3,7-8,18,27-29,32-33H,1-2,4-6,9-17H2,(H,30,34)(H4,25,26,31)Precursor 1 [M+H]⁺ 475.35035 Precursor 2 [M+2H]²⁺ 238.17881 Precursor 3 HDX 10 Precursor HDX 1 [M(D₁₀)+D]⁺ 486.41939 Precursor HDX 2 [M(D₁₀)+2D]²⁺ 244.21647 Precursor HDX 3 Rt 7.19 Rt HDX 5.69
Calculated MS/MS fragments # a b c ta z y tz 1 231.11280 213.10224 214.08626 248.13935 114.10257 97.07602 131.12912 2 288.17065 270.16009 271.14410 305.19720 171.16042 154.13387 188.18697 3 345.22850 327.21794 328.20195 362.25505 228.21827 211.19172 245.24482 4 416.30200 398.29144 399.27545 475.35035 285.27612 268.24957 302.30267
Additional MS/MS fragments m/z Annotation 146.06004 a’ 174.05495 a0
Recorded MS/MS spectra pdf Precursor Co-eluting Spider Source Author Data 475.35035 E. agrestis Fauna Laboratories Ltd., KAZ Y. M. Forster Data 238.17881 E. agrestis Fauna Laboratories Ltd., KAZ Y. M. Forster Data HDX E. agrestis Fauna Laboratories Ltd., KAZ Y. M. Forster Data 475.35035 P. luctuosa Fauna Laboratories Ltd., KAZ Y. M. Forster Data 238.17881 P. luctuosa Fauna Laboratories Ltd., KAZ Y. M. Forster Data HDX P. luctuosa Fauna Laboratories Ltd., KAZ Y. M. Forster
References Title Reference Spider Name Content Link Detection and characterization of natural polyamines by high-performance liquid chromatography-atmospheric pressure chemical ionization (electrospray ionization) mass spectroscopy S. Chesnov, L. Bigler, M. Hesse, Eur. J. Mass Spectrom. 2002, 8, 1-16 P. luctuosa PB 474 APCI-MS/MS Link Low molecular mass compounds in spider venom Y. M. Forster, S. Bienz, L. Bigler, 2020, in preparation div. Link
Spider species Spider species Family Discovered Eratigena agrestis Agelenidae 2020 / Y. M. Forster Pireneitega luctuosa Agelenidae 2002 / S. Chesnov