Navigation :
(Acyl)polyamines
-
2,4-(OH)₂-PhAc
-- 2,4-(OH)₂-PhAcAsn3(Me)43
-- 2,4-(OH)₂-PhAcAsn34
-- 2,4-(OH)₂-PhAcAsn343
-- 2,4-(OH)₂-PhAcAsn34ßAla3
-- 2,4-(OH)₂-PhAcAsn34ßAla4
-- 2,4-(OH)₂-PhAcAsn353
-- 2,4-(OH)₂-PhAcAsn4
-- 2,4-(OH)₂-PhAcAsn43
-- 2,4-(OH)₂-PhAcAsn433
-- 2,4-(OH)₂-PhAcAsn433Arg
-- 2,4-(OH)₂-PhAcAsn433Lys
-- 2,4-(OH)₂-PhAcAsn433ßAla4
-- 2,4-(OH)₂-PhAcAsn433ßAla4ßAla4
-- 2,4-(OH)₂-PhAcAsn43Arg
-- 2,4-(OH)₂-PhAcAsn43Lys
-- 2,4-(OH)₂-PhAcAsn43ßAla4
-- 2,4-(OH)₂-PhAcAsn4Lys(Me₂)3Arg⁺
-- 2,4-(OH)₂-PhAcAsn4Orn(NMe₂)
-- 2,4-(OH)₂-PhAcAsn4ßAla3Arg
-- 2,4-(OH)₂-PhAcAsn4ßAla4
-- 2,4-(OH)₂-PhAcAsn4ßAla43
-- 2,4-(OH)₂-PhAcAsn5
-- 2,4-(OH)₂-PhAcAsn53
-- 2,4-(OH)₂-PhAcAsn533
-- 2,4-(OH)₂-PhAcAsn533Arg
-- 2,4-(OH)₂-PhAcAsn5Gly4
-- 2,4-(OH)₂-PhAcAsn5Gly4ßAla4
-- 2,4-(OH)₂-PhAcAsn5Gly4ßAla4ßAla4
-- 2,4-(OH)₂-PhAcAsn5OrnArg
-- 2,4-(OH)₂-PhAcAsn5ßAla3
-- 2,4-(OH)₂-PhAcAsn5ßAla3Arg
-- 2,4-(OH)₂-PhAcAsn5ßAla3ßAla4ßAla4
-- 2,4-(OH)₂-PhAcAsn5ßAla4
-- 2,4-(OH)₂-PhAcAsn5ßAla43
-- 2,4-(OH)₂-PhAcAsn5ßAla43ßAla4
-- 2,4-(OH)₂-PhAcAsn5ßAla43ßAla4ßAla4
-- 2,4-(OH)₂-PhAcAsn5ßAla4AlaGlyArg
-- 2,4-(OH)₂-PhAcAsn5ßAla4Arg
-- 2,4-(OH)₂-PhAcAsn5ßAla4ßAla4
-- 2,4-(OH)₂-PhAcAsn5ßAla4ßAla4ßAla4
-- 2,4-(OH)₂-PhAcAsn5ßAla4ßAla4ßAla4ßAla4
-- 2,4-(OH)₂-PhAcOrnAsn5ßAla4ßAla4ßAla4
-
2,5-(OH)₂-Bz
-
4-OH-Bz
-
4-OH-IndAc
-
4-OH-PhAc
-
4-OH-PhLac
-
Ac
-
Bz
-
IndAc
-
IndLac
-
OH-OMe-Bz
-
PhAc
-
Phe
-
PhLac
-
Prop
-
Trp
-
Tyr
-
Unknown acylpolyamines
-
Free polyamines (PA)
Small compounds
Analytical tools
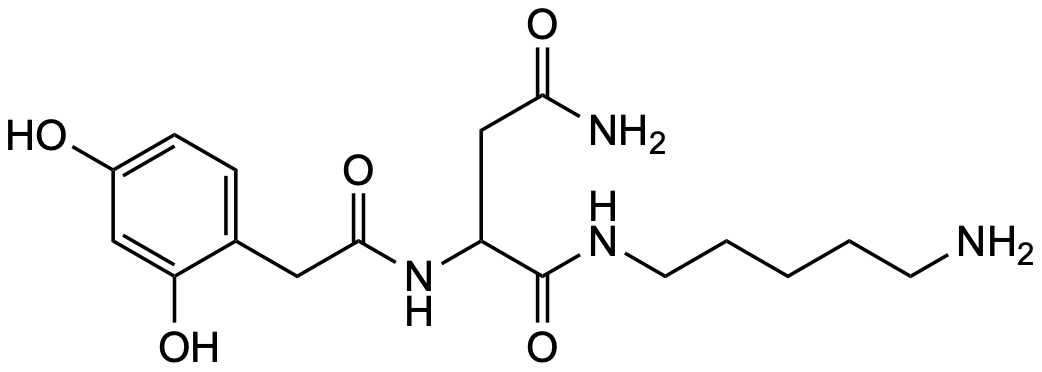
Contact us 2,4-(OH)₂-PhAcAsn5
General Description Name Value Level S-3 / C-1 Discovered 2020 / P. bistriata Synonym — Molecular formula C₁₇H₂₆N₄O₅ CAS 112111-38-3 SMILES O=C(CC1=CC=C(O)C=C1O)NC(CC(N)=O)C(NCCCCCN)=OInChI InChI=1S/C17H26N4O5/c18-6-2-1-3-7-20-17(26)13(10-15(19)24)21-16(25)8-11-4-5-12(22)9-14(11)23/h4-5,9,13,22-23H,1-3,6-8,10,18H2,(H2,19,24)(H,20,26)(H,21,25)Precursor 1 [M+H]⁺ 367.19760 Precursor 2 [M+2H]²⁺ 184.10244 Precursor 3 HDX 8 Precursor HDX 1 [M(D₈)+D]⁺ 376.25409 Precursor HDX 2 [M(D₈)+2D]²⁺ 189.13382 Precursor HDX 3 Rt 5.44 Rt HDX 4.50
Calculated MS/MS fragments # a b c ta z y tz 1 265.08190 247.07133 248.05535 282.10845 86.09643 69.06988 103.12297 2 350.17105 332.16048 333.14450 367.19760 200.13935 183.11280 217.16590
Additional MS/MS fragments Recorded MS/MS spectra pdf Precursor Co-eluting Spider Source Author Data 367.19760 P. bistriata Prof. Dr. Wagner Ferreira dos Santos, BRA Y. M. Forster Data HDX P. bistriata Prof. Dr. Wagner Ferreira dos Santos, BRA Y. M. Forster
References Title Reference Spider Name Content Link Synthesis of spider toxin (JSTX-3) and its analogs Y. Hashimoto, Y. Endo, K. Shudo, Y. Aramaki, N. Kawai, T. Nakajima, Tetrahedron Letters 1987, 28, 3511-3514 — compound 3 Synthesis, NMR, Activity-studies Link Newly synthesized analogues of the spider toxin block the crustacean glutamate receptor K. Shudo, Y. Endo, Y. Hashimoto, Y. Aramaki, T. Nakajima, N. Kawai, Neuroscience Research 1987, 5, 82-85 — C-1 Synthesis, Activity-studies Link Chemical structure of Joro spider toxin (JSTX) Y. Aramaki, T. Yasuhara, K. Schimazaki, N. Kawai, T. Nakajima, Biomed. Res. 1987, 8, 241 — Activity-studies Link Antropod toxins as leads for novel insecticides: An assessment of polyamine amides as glutamate antagonists I. S. Blagbrough, P. T. H. Brackley, M. Bruce, B. W. Bycroft, A. J. Mather, S. Millington, H. L. Sudan, P. N. R. Usherwood, Toxicon 1992, 30, 303-322 — Activity-studies Link Elucidation of the structure and synthesis of neuroprotective low molecular mass components of the Parawixia bistriata spider venom Y. M. Forster, J. L. Green, A. Khatiwada, J. L. Liberato, P. A. Narayana Reddy, J. M. Salvino, S. Bienz, L. Bigler, W. Ferreira dos Santos, A. C. K. Fontana, ACS Chem Neurosci., 2020 P. bistriata ESI-MS/MS Link
Spider species Spider species Family Discovered Parawixia bistriata Araneidae 2020 / Y. M. Forster